The Online Meeting was conducted by Dr. Carlos Eduardo dos Santos Ferreira, clinical pathologist, with a master’s and PhD in Medicine, Medical Manager at Einstein Hospital Israelita, Coordinator at the Laboratório Central da Escola Paulista de Medicina/Hospital de São Paulo, and Director of Accreditation and Quality at SBPC/ML.
The session covered the pre-analytical, analytical and post-analytical aspects of blood gas, electrolyte and metabolite testing, integrated with acid-base evaluation, as well as clinical and laboratory considerations.
With up-to-date content, the training provided a comprehensive overview of integrated blood gas analysis — from proper indication and collection of venous and arterial samples to transportation, storage, processing, quality control, and interpretation of results.
Questions & Answers
The following questions were not addressed during the Online Meeting.
Regarding harmonization/comparability between blood gas analyzers, could you indicate how to perform it considering the sample has time and air exposure limitations?
Process the samples quickly (within 30 minutes of collection) across the analyzers and evaluate the results, ensuring that routine sample syringes are not exposed to air. One option is to use external control samples for each analyzer as a measure of comparability.
How to perform harmonization if the blood gas analyzers are located in ICUs far apart within the hospital complex?
Same as the answer above.
What is the recommended frequency for harmonization?
At least once a year, while it is necessary to check whether accrediting institutions have modified the requirements.
What can increase PaO₂ in hospitalized patients who are not on mechanical ventilation?
They are receiving oxygen via mask or catheter.
Does the IQM process replace internal quality control?
No, they assess different aspects.
Regarding temperature correction in blood gas analysis, what is its importance?
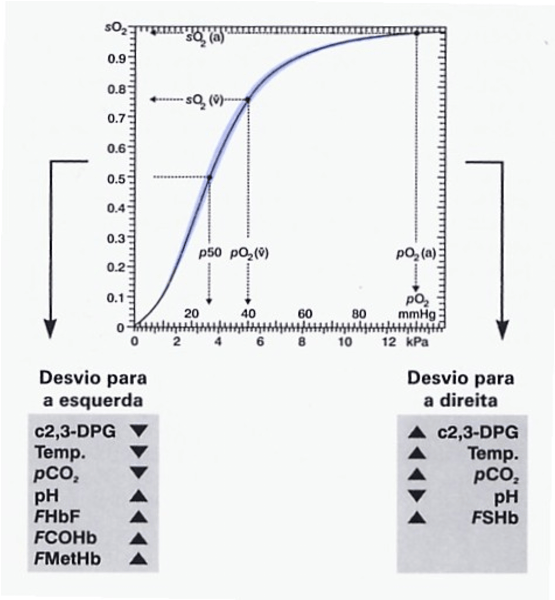
Correct values according to the sample temperature, as is done for atmospheric pressure correction (entered into the analyzer). Analyzers are standardized to 37 °C. In cases of hyperthermia or hypothermia, measured values of partial oxygen pressure, CO₂, and pH may not accurately reflect the in vivo physiological state, leading to incorrect clinical interpretations if not adjusted. For example, oxygen affinity decreases as temperature increases. Table below: oxygen dissociation curve.
Can blood gas be collected from the same puncture as routine lab tests (tubes)?
Yes. For venous blood samples. Use a heparin tube. Place it in the correct tube sequence. Closed system. Each collection system must be evaluated to determine whether it allows the use of a syringe with balanced heparin. Arterial blood—collected separately from an arterial site.
Is it possible to detect sample instability from blood gas values?
You may suspect it by evaluating clinical correlation. Evaluating only in the laboratory, it is not possible. Or values that require a new sample? Only with the laboratory, it is not possible.