The meeting was led by Dr. Regina Kiomi Takahira, a veterinarian graduated from the School of Veterinary Medicine and Animal Science at the University of São Paulo (FMVZ-USP), with residency, master’s, and doctoral degrees from the São Paulo State University “Júlio de Mesquita Filho” (FMVZ-Unesp) – Botucatu, where she is a Full Professor. She also serves as Vice President of the Brazilian Association of Veterinary Clinical Pathology (ABPCV).
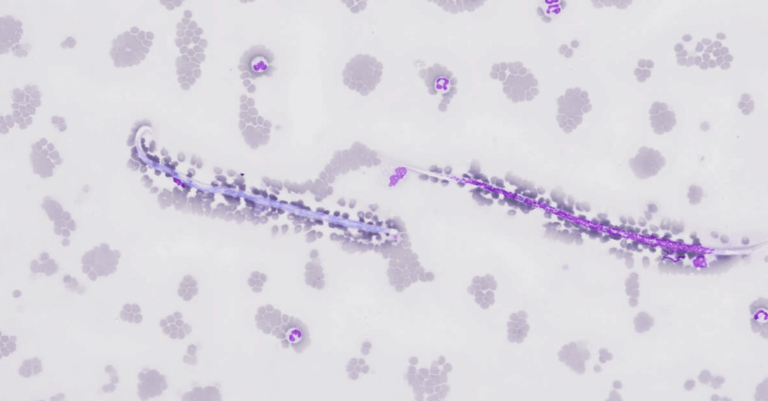
The presentation covered breed and species-specific predispositions, a key aspect for contextualizing laboratory findings. Breeds such as Dalmatians and Bulldogs, for example, have genetic characteristics that significantly alter their urine crystal profiles.
Additionally, the meeting emphasized the importance of clear and standardized technical communication among professionals involved in veterinary patient care, promoting consistency in reports and accuracy in clinical decision-making.
Questions & Answers
The following questions were not addressed during the Online Meeting.
First, it is important to ensure that the structures observed are indeed hemoglobin bodies and not other similar structures. The most appropriate method of confirmation is through fixation and staining of the sediment slide. Hemoglobin bodies exhibit color and texture similar to red blood cells but are smaller in size. They may appear alone or associated with red blood cells (hematuria). The description can be made qualitatively (simply noting their presence or absence) or quantitatively (indicating the presence of small, moderate, or large amounts of hemoglobin bodies, or using a plus system, such as rare hemoglobin bodies, +, ++, or +++, to be recorded in the observations section of the urinalysis report).
Yes. The urinary sediment evaluation protocol involves centrifuging a standardized volume of homogenized urine (usually 5 mL) and resuspending a fixed volume of sediment (0.5 mL) after discarding the supernatant. Centrifuging smaller volumes results in a lower concentration of formed elements (cells, casts, crystals, bacteria, and other structures) and, consequently, may lead to underestimation of the findings. On the other hand, smaller-volume samples are also more susceptible to pre-analytical factors, such as contamination from lower urinary or genital tract secretions, which can result in overestimation of leukocytes and bacteria, among other elements.
Hyaline casts can be observed in small amounts in the urine of healthy animals. Their formation may occur in situations of pre-glomerular proteinuria associated with increased filtration pressure, such as after intense physical activity. Other types of casts, however, are considered pathological and may be associated with tubular injury, among other alterations.
Bilirubin is a photosensitive molecule, unstable when exposed to visible light (especially ultraviolet light). Upon exposure, it undergoes photo-oxidative degradation, leading to the formation of colorless derivatives (biliverdin and other products). This degradation causes the bilirubin concentration to decrease artificially, resulting in falsely low or negative results. Studies show that bilirubin can undergo significant reductions within 30 minutes of light exposure, with a loss reaching 30–50% in 1 to 2 hours, depending on light intensity. For this reason, urine samples should be protected from light by wrapping the container in aluminum foil or storing the urine in amber tubes.