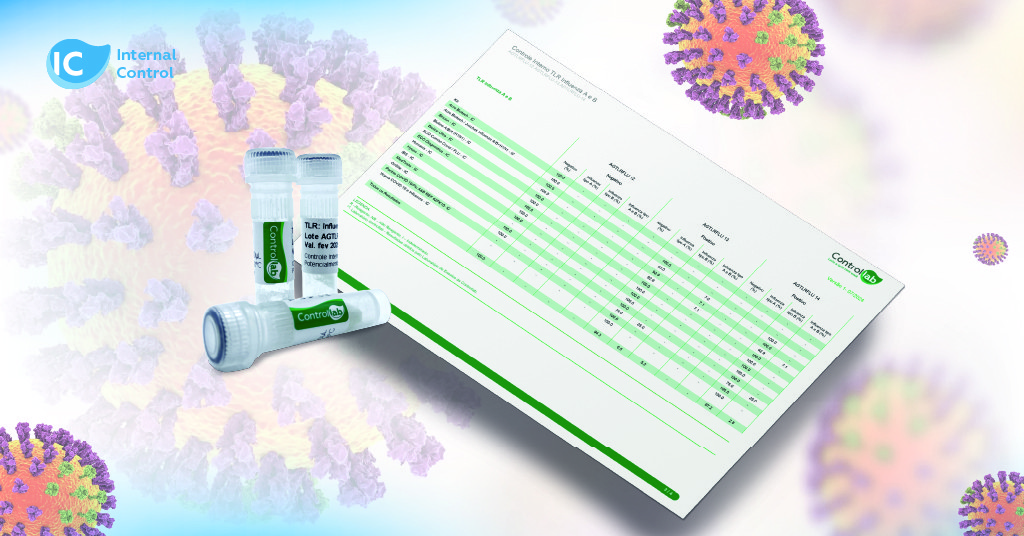
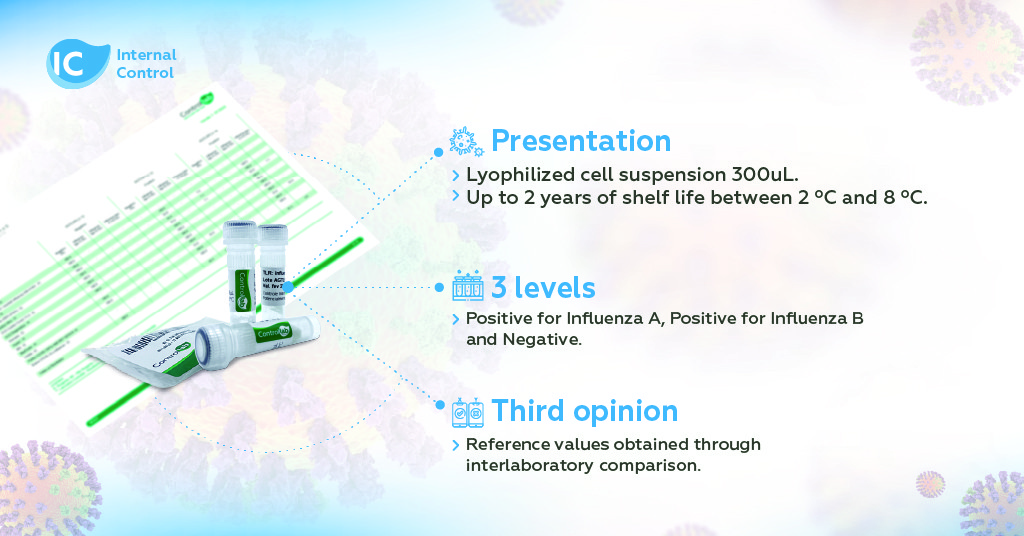
The Controllab Program includes three distinct levels (positive for Influenza A, positive for Influenza B, and negative), allowing the evaluation of interferences and cross-reactions in analytical systems.
Influenza viruses circulate year-round, intensifying in winter when people shelter from the cold in enclosed spaces, facilitating transmission. In Brazil, an increase in cases has been identified earlier than expected.
This anticipation, combined with other circulating diseases, has required rapid and accurate diagnostics to prevent the worsening of illnesses and the overloading of the healthcare system, avoiding self-medication, worsening of symptoms, hospitalizations, or deaths.
Reliability in analytical routines
In this scenario, clinical analysis establishments need to have their routines monitored to prevent incorrect reports from contributing to the lack of adequate diagnoses.
At the forefront of contributing to the improvement of the quality of healthcare services offered to the population, Controllab provides an additional control to help establishments demonstrate the reliability of their routines: POCT Influenza A and B.
The control is composed of three distinct levels:
- positive specific for Influenza A;
- positive specific for Influenza B;
- negative.
This approach allows for the evaluation of potential interferences and cross-reactions in analytical systems. Additionally, the control includes reference values obtained through interlaboratory comparison, reflecting the reality of various laboratories using different POCT systems.
The quantitative and qualitative data are grouped by POCT device, presenting the performance of these systems individually.
External and internal controls
Controllab provides external and internal controls that raise the standard of analytical routines. Controllab’s internal control materials are unbiased (third-party opinion) and detect variations not perceived in the controls provided by the manufacturers of the analytical system.
In addition to POCT Influenza A and B, Controllab also offers internal controls for POCT Respiratory Syncytial Virus (RSV) and POCT Coronavirus (SARS-CoV2), ensuring reliable monitoring of respiratory virus routines.
Do you want recognition for excellence in your flu virus reports?
Speak now with a Controllab specialist and boost your establishment’s reputation and your audience’s satisfaction.