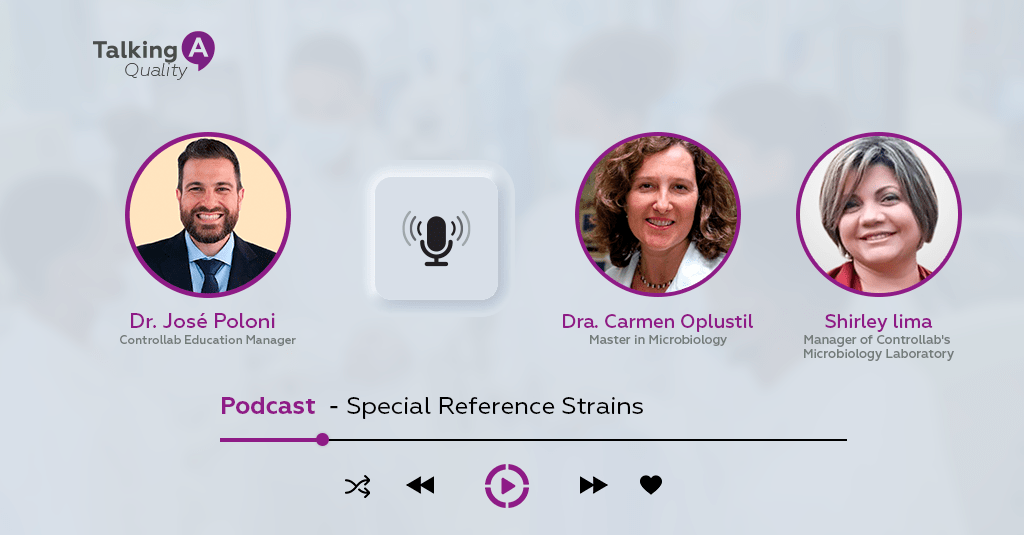
In podcast format, “Talking A” received experts who highlighted the use of strains in the quality control processes of diagnoses.
Controllab’s educational manager, José Antonio Tesser Poloni, interviewed Dr. Carmen Paz Oplustil, who holds a master’s degree in microbiology from the Institute of Biomedical Sciences at the University of São Paulo (ICB-USP), and Shirley Lima, manager of Controllab’s microbiology laboratory , in yet another edition of the Talking About Quality. The specialists highlighted the importance of reference strains for the day-to-day activities of the microbiology sector.
Strains and quality control
Dr. Carmen, who is also the director of Format Clinical Brazil and Chile, is the author of the online course “Maintenance and use of Reference Strains (ATCC) without complications”.
During the interview, the specialist pointed out that this free course, available in Portuguese and Spanish, aims to inform and facilitate the activity of laboratory professionals, providing clear guidelines on the handling and use of reference strains during quality control processes.
An important highlight is the application of these strains in the antibiogram test, which evaluates the patterns of resistance or susceptibility of a bacterium to various antibiotics and chemotherapy.
Dr. Carmen also emphasized that the reference strains can be considered as a calibration standard. They should be used whenever there is a need to prove the proper functioning of a test or test, providing reliability to the diagnostic results.
Strains recognized as Certified Reference Material (CRM)
The manager of Controllab’s microbiology laboratory, Shirley Lima, emphasized that the licensing granted to the company was a great advance for laboratories in the clinical and environmental areas of Brazil and Latin America.
Controllab sends reference strains accompanied by certificates of analysis. They are materials for immediate delivery and in the quantity defined by the laboratory, based on the selected microorganisms. These certified strains provide more confidence and credibility in quality control, facilitating compliance with standards, audits, accreditations and inspections.
Click here and check out the new approach of Talking About Quality! Enjoy and subscribe to Controllab’s YouTube channel to have first-hand access to the educational content produced by the company.