Healthcare institutions have the opportunity to highlight their commitment to continuous improvement and patient safety, earning even more public trust.
Metricare is a comparative indicator management platform that facilitates decision-making in healthcare and wellness companies. Developed by Controllab and endorsed by the Brazilian Society of Clinical Pathology / Laboratory Medicine in its laboratory module, the platform enables organizations to enhance their management by monitoring performance indicators and benchmarking against the market.
Comparative indicator management in practice
Metricare provides participants with essential data for laboratory management, such as sample recollection rates, team productivity, result delivery delays, incorrect reports, average ticket size, performance in Proficiency Testing, and many other indicators – check out the full list.
For example, let’s look at some data on the Result Delay Indicator:
- In 2023, half of the laboratories recorded 12 delayed results per 1,000 patients.
- Laboratories with lower performance had 23 delays per 1,000 patients, while those with higher performance recorded only 5 delays per 1,000 patients.
Result Delay Index per 1,000 patients

Source: "Result Delay (non-inpatient)" indicator, comparative group performing between 50,000 and 300,000 tests/month, Metricare 2023.
The result delay indicator reflects a laboratory’s efficiency in meeting deadlines and its influence on physician and patient satisfaction. Delays can compromise patient safety and the lab’s competitiveness, while shorter turnaround times demand more agile processes. Setting deadlines strategically is crucial to balancing quality, efficiency, and sustainability.
How could laboratories that recorded 12 or 23 delays per 1,000 patients reassess their internal targets without market data? Without this information, how would they understand how much they still need to improve to become more competitive and attractive to their audience?
Continuously tracking the benchmarking results of laboratory indicators enables more accurate decision-making, helping define goals and implement strategic actions. This leads to continuous improvements in processes and patient safety—a competitive advantage for laboratories that invest in quality management.
Metricare participants who have earned the Participation Certificate can now showcase this commitment to the public. Discover the certified laboratories in 2024 and see how they stand out in the market.

What does the certificate represent for your audience?
Achieving the Metricare Participation Certificate reflects a laboratory’s commitment to best management practices, driving continuous improvements that benefit patients, healthcare professionals, managers, and the entire healthcare ecosystem.
The certificate represents:
- Commitment to high standards, promoting practices that reduce risks and increase result reliability.
- Contribution to continuous quality improvement in the sector, ensuring greater sustainability in healthcare.
- Performance indicator-based management, aligned with NBR ISO 15189.
How to use the certificate to strengthen your reputation and highlight your laboratory
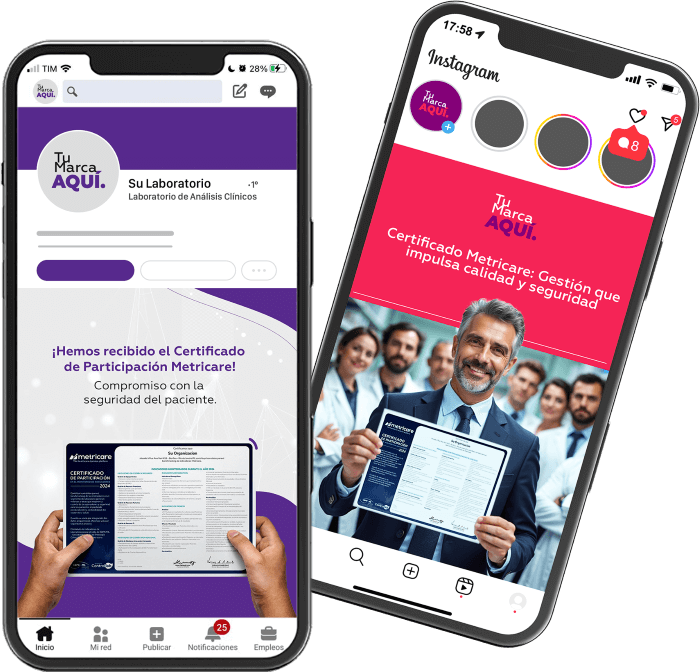
Sharing this achievement enhances your laboratory’s image. By promoting it on your website, social media, and other institutional channels, you:
- Demonstrate your commitment to patient safety.
- Showcase compliance and efficient management practices.
- Strengthen public trust.
- Differentiate your laboratory in the market.
To make this easier, Controllab provides an exclusive page with suggestions on how to maximize the impact of your certification in public perception.
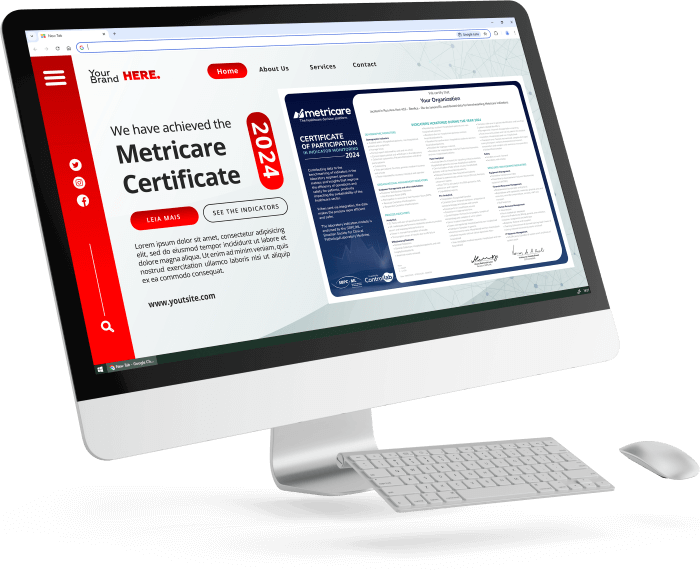
How does the certificate promotion page help your laboratory?
This page serves as a guide for your marketing and communication team, offering:
- Guidelines on how to highlight strategic information about your certificate.
- Text and post suggestions, making communication on social media and other media platforms easier.
Start showcasing your competitive advantage now
Take advantage of the exclusive page with practical suggestions to highlight your Metricare Participation Certificate. Explore this resource and increase your visibility in the market!
Your audience needs to know about your commitment!
Not yet part of Metricare?
Gain access to strategic data that drive smart decisions, highlight your commitment to continuous improvement and patient safety, and strengthen your laboratory’s reputation.
Start reporting data now and position your institution among the leading laboratories in quality management, contributing to sector improvement.
Speak with our specialists and discover how your laboratory can benefit from comparative indicator management. Contact us via WhatsApp: +55 21 98258-0074!




