Background
Organizations employ performance monitoring systems, with performance indicators as a widely adopted practice. In Brazil, the Metricare Platform facilitates comparative analysis for laboratory indicators across 17 countries, encompassing 380 registered laboratories. The program aims to empower laboratories for self-identification of improvement opportunities and foster positive marketwide performance evolution.
Aim
This study aims to compare laboratory performance metrics within the indicator’s platform, focusing on critical indicators related to quality, safety, and outcomes.
Methods
The Metricare platform, aligned with International Federation of Clinical Chemistry (IFCC) harmonization, receives monthly data submissions from participating laboratories. Five key indicators (Performance in Proficiency Tests; Incorrect reports; Failure to communicate critical results; Delay in results for outpatients; and Sample recollection) were selected to assess performance evolution. The median of the comparative group for each indicator in the global sampling for 2018 and 2022 was analyzed. Performance results for both years, expressed in the sigma (s) metric for the 50th percentile, were compared to identify statistically significant performance differences.
Results
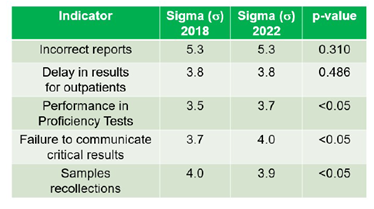
Two indicators did not show statistically significant differences between 2018 and 2022 (Incorrect reports, s 5.3, p=0.310; Delay in results for outpatients, s 3.8, p=0.486). Two others showed performance improvement (Performance in Proficiency Tests, s 3.5 to 3.7, p<0.05; Failure to communicate critical results, s 3.7 to 4.0, p<0.05).
One of the indicators, Sample recollections, showed a moderate decrease in performance (s 4.0 to 3.9, p<0.05) (Table 1). These performance levels are aligned with scientific publications that address clinical laboratory performance.
Conclusions
Although no drastic changes were observed, enhancements in laboratory performance for two indicators were identified. These positive trends contribute to patient safety. The consistent data coherence underscores the reliability of the indicator platform. The finding of improved performance among participants for some indicators suggests the platform’s contribution in guiding laboratory improvements and promoting higher-quality and safer laboratory services for patients.
References
Shcolnik W, Berlitz F, Galoro C A O, Biasoli V, Lopes R, Jerônimo D, Balli L B, Bernardes L. Brazilian laboratory indicators benchmarking program: three-year experience on pre-analytical quality indicators. Diag 2021; 8: 257-268.
Sciacovelli, L., Padoan, A., Aita, A., Basso, D., & Plebani, M. (2023). Quality indicators in laboratory medicine: stateof- the-art, quality specifications and future strategies. Clinical chemistry and laboratory medicine, 61(4), 688–695.
Arumalla VK, Chelliah S, Madhubala V. Evaluation of preanalytical phase performance of emergency laboratory byharmonized quality indicators and six sigma. J Med Sci Res. 2021; 9(2):64-70.