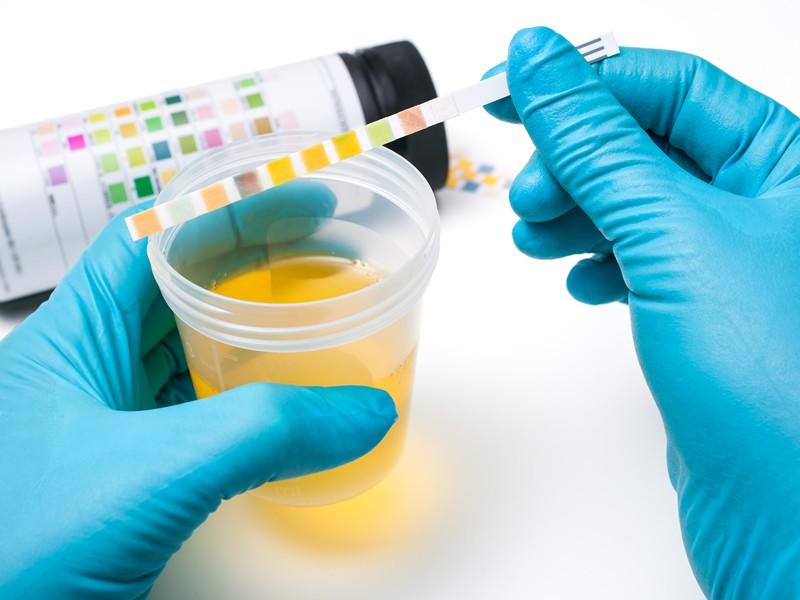
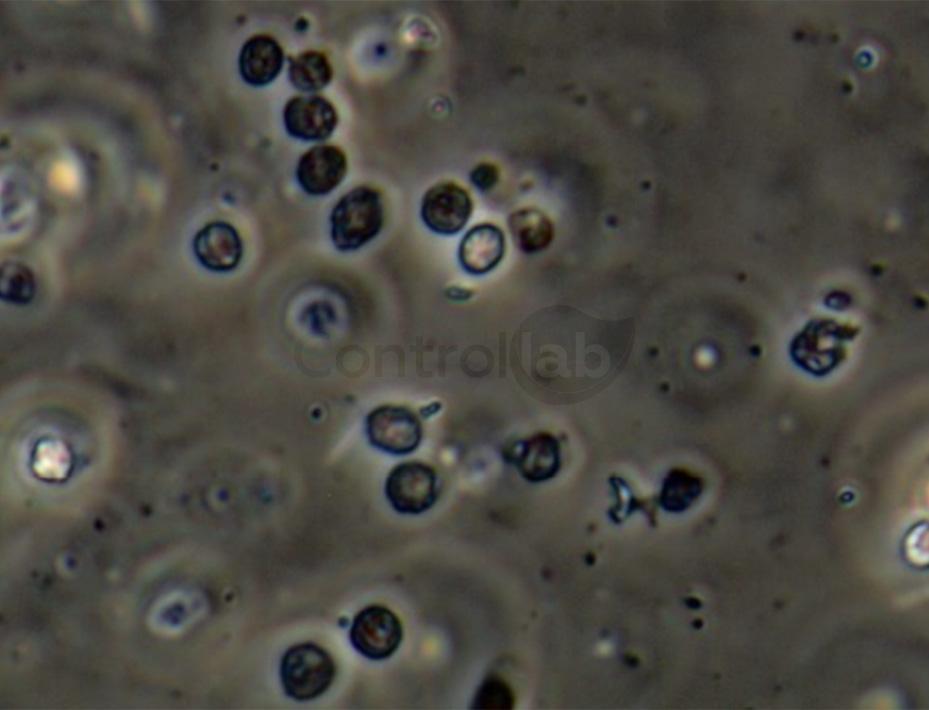
Urine testing is capable of diagnosing several diseases and has had great evolution in recent years through new technologies
Urinalysis includes evaluations that help in the diagnosis of kidney and urogenital tract diseases, in addition to liver, hemolytic and carbohydrate metabolism disorders. With simple and low-cost exams, the area has grown in interest and is increasingly valued.
To issue assertive reports of urine tests, the routine requires well-trained professionals, adequate equipment and the application of standardized methodologies. In recent years, these tests have evolved greatly thanks to the availability of new technologies for evaluating samples, with new parameters for the physical-chemical examination and high-quality resources for the identification of urinary particles, such as Flow Cytometry.
Importantly, in addition to Cytometry, cuvette-based microscopy and automated intelligent microscopy are also used to identify detectable elements in urinary sediment.
A particularly interesting feature for the identification of kidney-related clinical conditions is the possibility offered by some manufacturers to identify the albumin/creatinine ratio and the protein/creatinine ratio via a test strip.
The development and greater offer of automated equipment optimize the workflow of Urinalysis routines, allowing a quick and safe release of negative samples and the correct signaling for reviewing samples with pathological findings.
This process evolution is accompanied by quality control providers, with specific programs for automated routines, as well as for manual techniques. For example, the External Control for Research on Erythrocyte Dysmosrphism, made available in a pioneering way by Controllab.
This research is a relevant test for the identification of hematuria of glomerular origin. Participation in continuous External Control allows for the evaluation and continued education of the professionals who perform this test.
Process Control
Microscopic analysis is profoundly impacted by the knowledge of the professional who performs the evaluation of the samples, the available microscopy applications (phase contrast filters and polarized light which are the gold standard resources for the analysis of urinary sediment) and compliance with the protocol standardized for the test.
Therefore, quality control becomes essential to eliminate or minimize these impacts. The continuous evaluation of the results provided by the equipment and the human skills applied in the analysis of the samples, ensure the correct identification of the elements present in the urine.
“Microscopic analysis is, without a doubt, the point that requires the longest time to achieve an adequate level of training for professionals. Depending on the type of samples that make up the Urinalysis routine, it can be quite challenging to achieve an appropriate qualification. For example, centers professionals that treat a large number of patients with kidney disease and transplant recipients will require a greater number of hours of training than centers professionals that work with outpatient samples”, explains Dr. José Poloni – professor at the Escola de Saúde da Unisinos and scientific advisor of Controllab Urinalysis Program.
The external and internal control tools are important allies of efficient routines and, consequently, the emission of reliable results. There are also national and international regulations that collaborate in these processes. For example, ABNT is revising the standard for urine testing and an updated version will soon be available. CLSI has a specific document with guidelines for urinalysis and there is also the European Guide for Urinalysis.
The Brazilian Society of Clinical Pathology and Laboratory Medicine (SBPC/ML) has available in its digital library a book with “Recommendations for the Urine Examination”. Controllab also provides free books on Analytical Phase Management, including a chapter dedicated to “Process Control in Urinalysis”.
The challenges
Standardizing the procedures for performing the tests, with the adoption of protocols that follow national or international regulations among the professionals who work in these analyses, is a necessity of Urinalysis, aiming at standardizing the way of working and improving the process. In this sense, in Brazil, a recent initiative was the subject of an article, highlighting the importance of distinguishing epithelial cells in routine.
Additionally, it is essential that there is investment in human and technological resources for the sector, such as basic and continuous training of professionals and in microscopy, which plays a fundamental role in identifying the elements of the sediment. Finally, it is crucial to understand the strengths and weaknesses of the exam, making proper use of this test.
Controllab Urinalysis Program
Controllab is among the largest providers of laboratory quality control in the world, with more than 3,500 tests available in the portfolio. For Urinalysis, there is a comprehensive scope of exams that meet the human and veterinary matrices.
Controllab’s program includes all the variables that need to be evaluated in the routine of urine tests, including the analyses:
- Physicochemical;
- Quantitative microscopic for counting leukocytes and erythrocytes;
- Qualitative microscopic for visualization of urinary sediment;
- Clinical cases simulating everyday situations with information from the physicochemical examination and high-quality photographs of the elements of the urinary sediment in each case.
The program uses lyophilized samples to ensure stability during transport, virtual slides with the possibility of approximation and adjustment of focus that guarantee the standardization of the material, the diversity of cases and the inclusion of rare frames. Images of structures for identification are also contemplated, in addition to photographs of fresh sediment, in phase contrast, polarized light or stained by Sternheimer-Malbin.
Quality control is modularized to suit different routines. It instills confidence and credibility in analyses, enhances professional knowledge and promotes excellence in urine laboratory testing.
Discover the Controllab Urinalysis Program.