Background - Aim
The indirect immunofluorescence assay on HEp-2 cells (HEp-2 IFA) remains the gold standard for autoantibody screening, offering insights into potential autoantibodies and guiding further testing based on specific clinically relevant patterns. To promote harmonization in testing and reporting, the Brazilian Consensus on Antinuclear Antibodies (BCA HEp-2), established in 2000, and the International Consensus on ANA Patterns (ICAP), initiated in 2014, collaborate to standardize HEp-2 IFA pattern nomenclature and definitions. This study aims to assess Brazilian laboratories’ ability to classify common HEp-2 IFA patterns following BCA and ICAP guidelines.
Methods
In phase one, serum samples exhibiting six competent-level patterns were sent to 64 laboratories, and accuracy rates were calculated based on the correct identification of the expected patterns. Phase two evaluated image recognition accuracy for 22 additional patterns, with accuracy expressed as percentages. All patterns were reported using the alphanumeric code (AC) defined by ICAP (www.anapatterns.org).
Results
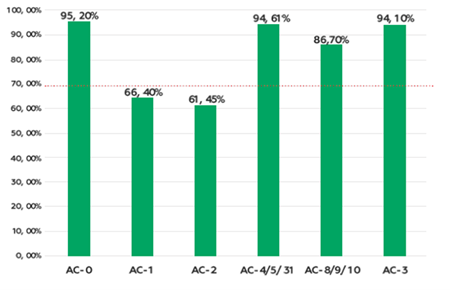
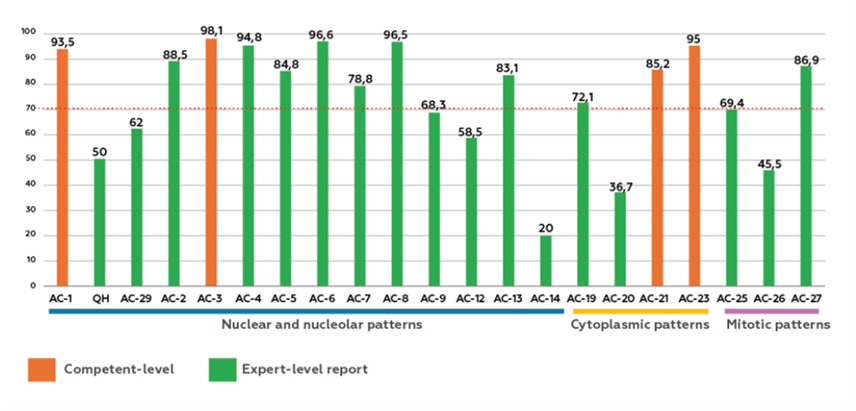
Negative HEp-2 IFA samples (AC-0) were correctly identified by 95.2% of participants. Positive sample accuracy rates varied: AC-1 (66.4%), AC-2/30 (61.4%), AC-3 (94.1%), AC-4/5/31 (94.6%), and AC-8/9/10 (86.7%) (Figure 1). Expert-level image recognition averaged 69.7%, with specific rates of 77.1% (nuclear), 82.4% (nucleolar), 72.2% (cytoplasmic), and 67.3% (mitotic). Accuracy for individual patterns ranged widely: AC-1 (93.5%), quasi-homogeneous (QH) nuclear (50.0%), AC-2 (88.5%), AC-3 (98.1%), AC-4 (94.8%), AC-5 (84.8%), AC-6 (96.6%), AC-7 (78.8%), AC-8 (96.5%), AC-9 (68.3%), AC-12 (58.5%), AC-13 (83.1%), AC-14 (20.0%), AC-19 (72.1%), AC-20 (36.7%), AC-21 (85.2%), AC-23 (95.0%), AC-25 (69.4%), AC-26 (45.5%), AC-27 (86.9%), and AC-29 (62.0%) (Figure 2).
Conclusions
Laboratories performed well with AC-0 and achieved high accuracy for most competent-level patterns. However, nuclear patterns with positive metaphase plates (AC-1, AC-2, QH, AC-29) presented significant challenges. Cytoplasmic and mitotic patterns were less consistently recognized than nuclear and nucleolar patterns. These results highlight the need for continued education to enhance pattern recognition, aligning with BCA and ICAP recommendations.
References
ANDRADE, Luis EC, Werner Klotz, Manfred Herold, eta l. Reflecting on a decade of the international consensus on ANA patterns (ICAP): Accomplishments and challenges from the perspective of the 7th ICAP workshop, Autoimmunity Reviews, 2024, 103608, ISSN 1568-9972, https://doi.org/10.1016/j.autrev.2024.103608.
CRUVINEL, Wilson M., ANDRADE, Luiz EC, DELLAVANCE, Alessandra et al. VI Brazilian consensus guidelines for detection of anti-cell autoantibodies on Hep-2. Adv Rheumatol, v. 62, n. 34, 2022
