The procedure brings fundamental information to the clinician, contributing significantly to the diagnosis and follow-up of pathologies related to infertility
The Spermogram is a laboratory test capable of analyzing male fertility potential. It serves to investigate the physical-chemical and microscopic parameters of human semen.
The Spermogram is the first test to be requested when dealing with marital infertility. The procedure brings important information to the clinician, such as concentration, motility, vitality and sperm morphology, contributing significantly to the diagnosis and monitoring of pathologies related to infertility.
The exam presents subjectivities. This characteristic fundamentally requires a well-monitored environment with frequent training, to avoid inter-examiner analytical variations.
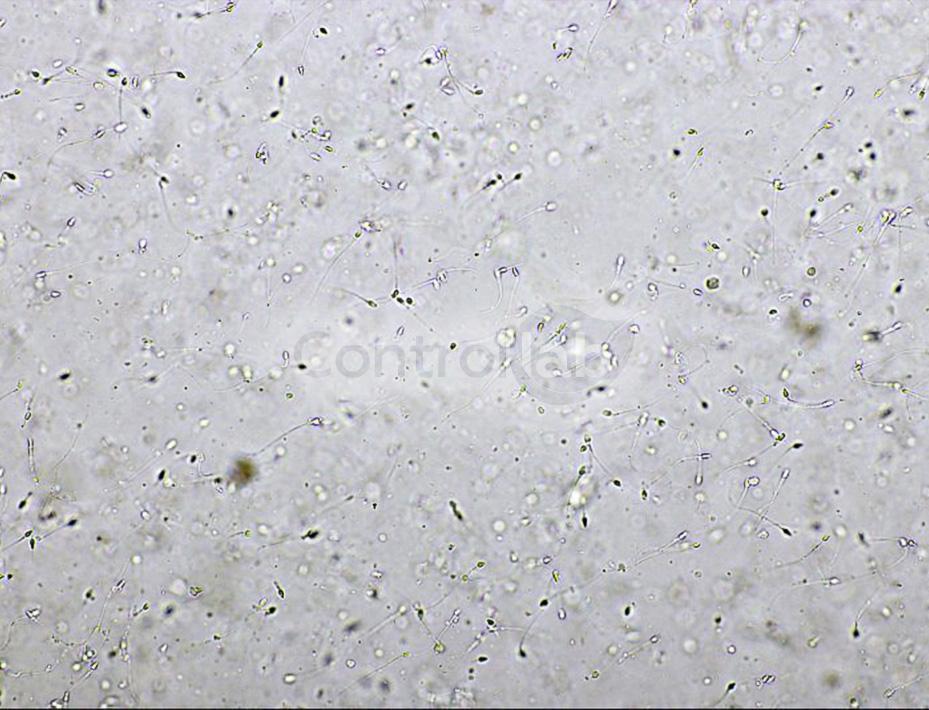
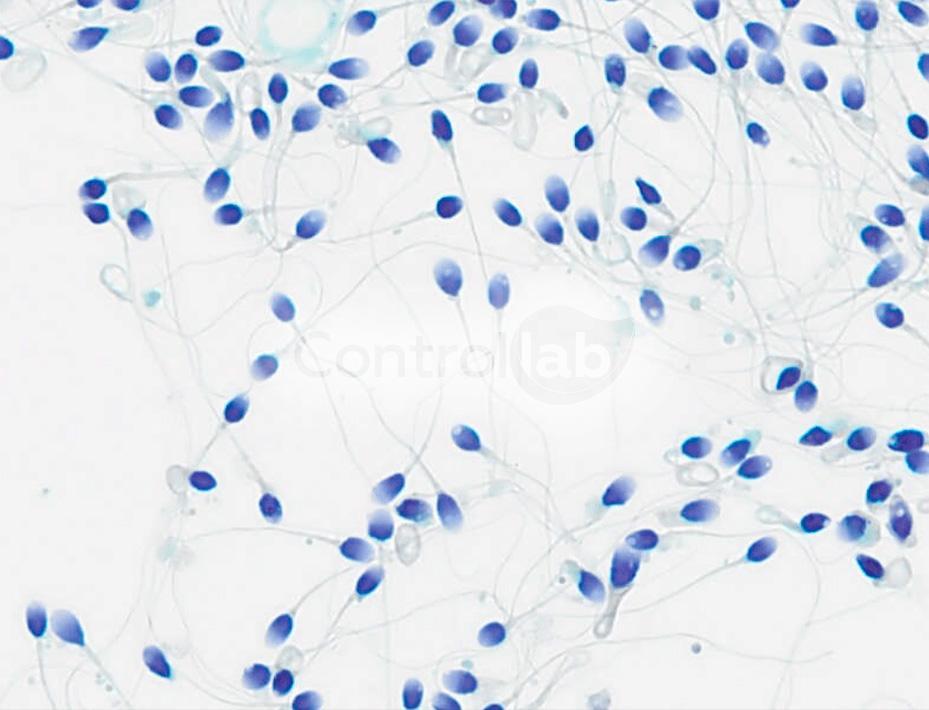
The microscopic analysis is one of the steps of the Spermogram. In it, there is the morphological evaluation of the semen. The strict sperm morphology has proved to be a strong ally in the search for a diagnosis of infertility, in cases where the other parameters analyzed are within the normal range.
The depth counting chambers have stood out to improve the performance of analysis, as well as the automation of the Spermogram has become sophisticated, improving the cost-effectiveness of implementation in laboratories and significantly reducing the subjectivity of the exam.
Process control
The quality of the preparations must be observed as one of the characteristics that impact the control of the process. It is necessary that laboratories continually invest in the training and evaluation of professionals, to avoid interexaminer analytical variations.
In this sense, the use of quality control is essential. It assists in the training and evaluation of the team, points out analytical variations, allows the identification of failures and makes it possible to take corrective or preventive actions. By continuously applying quality control in the routine, the laboratory has an easier time integrating and standardizing the examiners’ analyses, adding more reliability into the analytical processes.
Despite the difficulties of laboratories in obtaining control samples for Spermogram, it is possible to find a quality control provider with a comprehensive scope of analysis. At Controllab, for example, it is covered from chemical analyzes to sperm motility and morphology. These last two tests, if not well standardized in the routine of laboratories, impact on deviations in results in the reports.
According to Dr. Orildo dos Santos Pereira, author of the book Spermogram – Manual of Bench and Atlas, the exam still has some major challenges, such as reducing pre-analytical and analytical variations with correct standardization of procedures.
Thinking about these challenges, Dr. Orildo wrote the book seeking to provide information in an objective way, providing professionals with basic conditions for carrying out the examination and interpreting the patient’s clinical status in correlation with their reproductive capacity. All information is based on the WHO Manual.
The author adds that in September 2021, the 6th edition of the WHO Manual was published, which is the international reference for Spermogram analysis and must be followed by all laboratories that perform the test.
Controllab’s Quality Control Programs
Controllab uses the technology of digitized microscopy to enable some modules, presenting excellent quality in videos, images and virtual slides. This technology allows the participant to have an experience very close to the Spermogram routine and consequently more security in the release of the results.
Check the available programs:
Module | Tests | Service |
Sperm Biochemical | Citric acid Fructose Zinc | PT |
Sperm Cell Count and Vitality | Sperm Concentration Sperm Count Post-Vasectomy Total Sperm (THO) Live Sperm | PT |
Sperm Cell Count by Automation | Sperm Cell Count by Automation | PT, IC |
Sperm Motility | Percentage Sperm Motility | PT |
Sperm Morphology | Normal Sperm Peroxidase Positive Cell Count Relevant Defects Germinated Elements RBC’s Leukocytes | PT |
EA = Ensayo de Aptitud, también conocido como Control Externo.
CI = Control Interno
Para obtener más información, acceda al catálogo de Controllab.